 
Therefore, a neutron is given a relative charge of zero. A neutron has approximately the same mass as a proton, but it does not have a charge. These charges are equal in size but have opposite signs. The relative charge assigned to an electron is negative one, and the relative charge assigned to a proton is positive one. What differs about their charges is that the sign is opposite. However, the size or magnitude of the charge of an electron and a proton is equal. When comparing an electron to a proton, a proton is far more massive. The protons are positively charged, and the neutrons are neutral and do not have a charge. The nucleus of the atom contains a combination of protons and neutrons. Electrons are the negatively charged subatomic particles in the atom. The space outside the nucleus but still within the atom, often referred to as the electron cloud, is where the electrons are found. The dense core in the center of an atom is called the nucleus. They have no net charge, even though some of the subatomic particles inside the atoms do have charges. 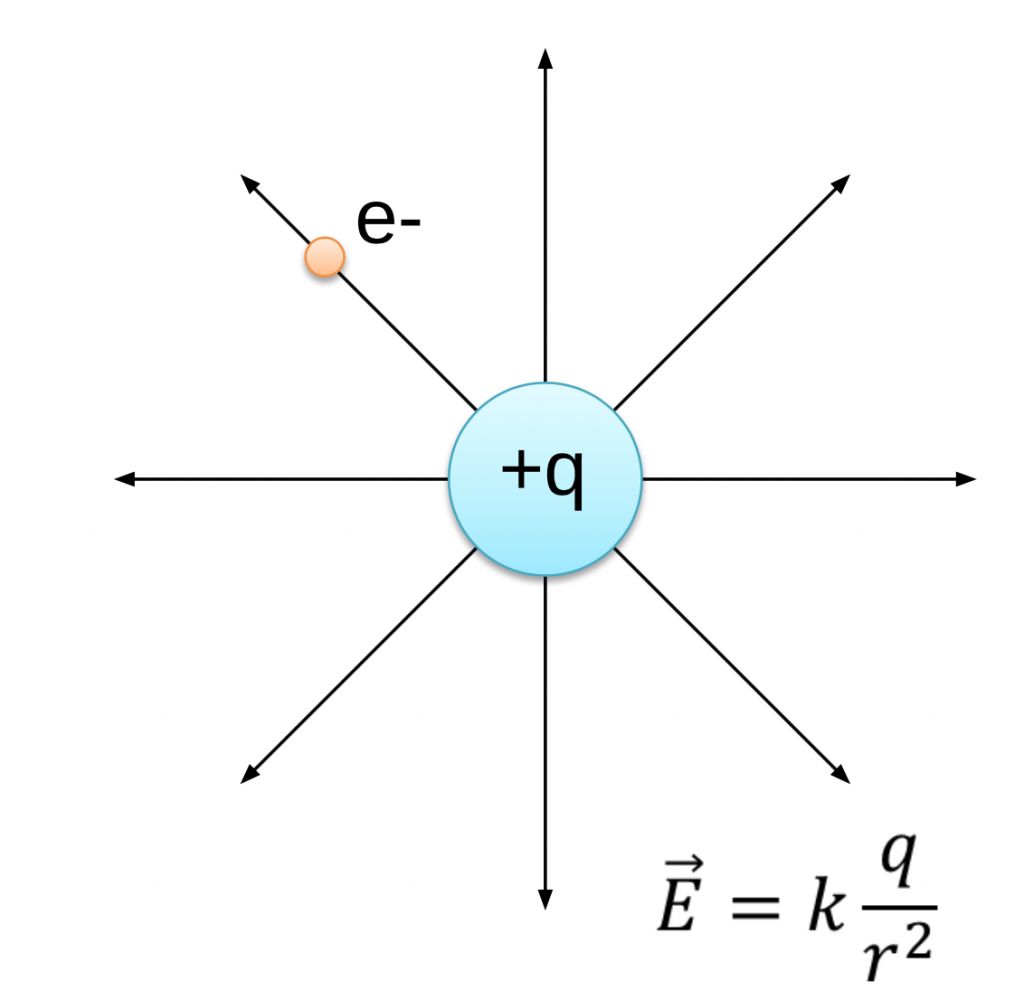
Atoms of elements are electrically neutral. Giving subatomic particles relative charges is very useful because it can help chemists keep track of the net charge of atoms and ions. The word neutral is typically used to describe particles with no charge. A particle can have a positive electrostatic charge, a negative electrostatic charge, or have no charge at all. 
When speaking of the charge on a subatomic particle at rest, we call it an electrostatic charge. Instead, we need to find out how the charges of the subatomic particles compare to one another. We are not concerned with the exact amount of charge carried by a subatomic particle, which could be measured. To solve this problem, we’ll need to determine the relative charges on these three different subatomic particles. To begin, it’s important to recognize that protons, neutrons, and electrons are three different types of subatomic particles found inside the atoms of chemical elements. Under some circumstances, protons and electrons can be converted to other particles in certain nuclear reactions, but in doing so, the net charge for the reactions is zero.What are the relative charges of a proton, a neutron, and an electron? Note: While it is a good model to think of conservation as an inability to increase or decrease the total number of protons and electrons, it technically isn't 100% accurate. These are referred to as the elementary charge. To give a brief quantitative overview of electric charge, the unit for charge is the Coulomb, denoted by "C". If a baseball is thrown upwards at an initial kinetic energy, #E_k#, the gravitational potential energy, #E_"PE"#, will be equal to #E_k#. We usually use this principle in physics when we equate the initial energy of an event to the final energy of an event. Another common conservation principle is energy. Here, it is the conservation of mass that is concerned. When you balance chemical equations, you are ensuring that the total number of atoms remain constant throughout the reaction. 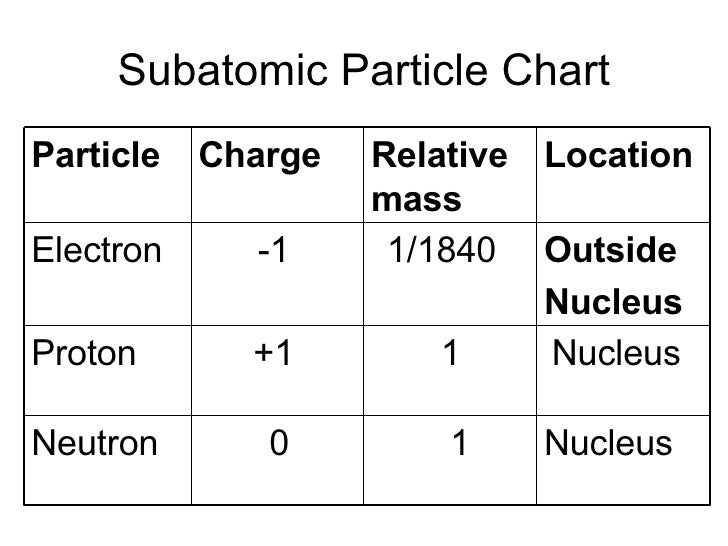
One way to think about conserved properties is that the total number of protons and electrons in the universe is constant (see Note below).Ĭonservation is a common theme in chemistry and physics. Since protons and electrons are the carriers of positive and negative charges, and they cannot be created or destroyed, electric charges cannot be created or destroyed. Simply put, protons and electrons cannot be created or destroyed. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
